The salts have some common characteristics, among them, the conduction of electric current when they are in solution and salty flavor. Salts are ionic compounds, have a salty taste, and are solid.
Estimated Reading Time: 3 minutes
Salts characteristics:
1 – Conduct electric current when they are in solution.
2 – Salts taste salty.
3 – The salts react with acids, with hydroxides, with other salts, and with metals.
4 – When reacting with an acid, they give rise to another salt and another acid if the acid formed is more volatile than that used in the reaction.
5 – When they react with hydroxide, they give rise to another salt and another hydroxide if the hydroxide formed is less soluble than that used in the reaction.
6 – If they react with other salts, they give rise to two new salts if one of them is less soluble than the reagents.
7 – And, finally, when they react with metal, they give rise to a new salt and a new metal if the metal is more reactive than the metal displaced in the reaction.
Main salts and their uses:
Sodium bicarbonate (NaHCO3) -> It is used in medicines that act as stomach antacids. It is also used as yeast in the manufacture of breads, cakes, etc., since it releases heated carbon dioxide, which allows the dough to grow. It is also used to manufacture foam for fire extinguishers.
Calcium Carbonate (CaCO3) –> Part of the marble, it is used in the making of floors, sinks, etc. Calcium carbonate (limestone) is also used in the manufacture of common glass and cement.
Calcium Sulfate (CaSO4) –> It is a salt used in the manufacture of chalk and porcelain plaster.
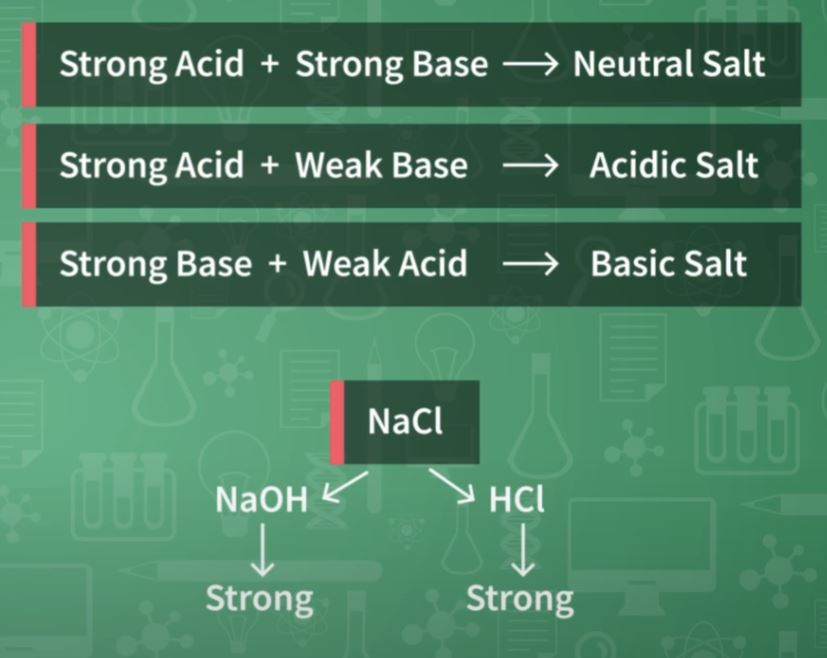
Sodium Chloride (NaCl) –> This salt is used intensely in food and also in the conservation of certain foods; in addition, it is one of the components of home serum, used to combat dehydration. In the kitchen salt, besides sodium chloride there is a small amount of sodium iodide (Nal) and potassium (Kl).
Sodium Fluoride (NaF) –> It is a salt used in the fluoridation of drinking water and as an anti-carious product, in the manufacture of toothpaste.
Sodium Nitrate (NaNO 3) –> Known as Chilean saltpetre, this salt is one of the most common nitrogen fertilizers.
Related posts:
